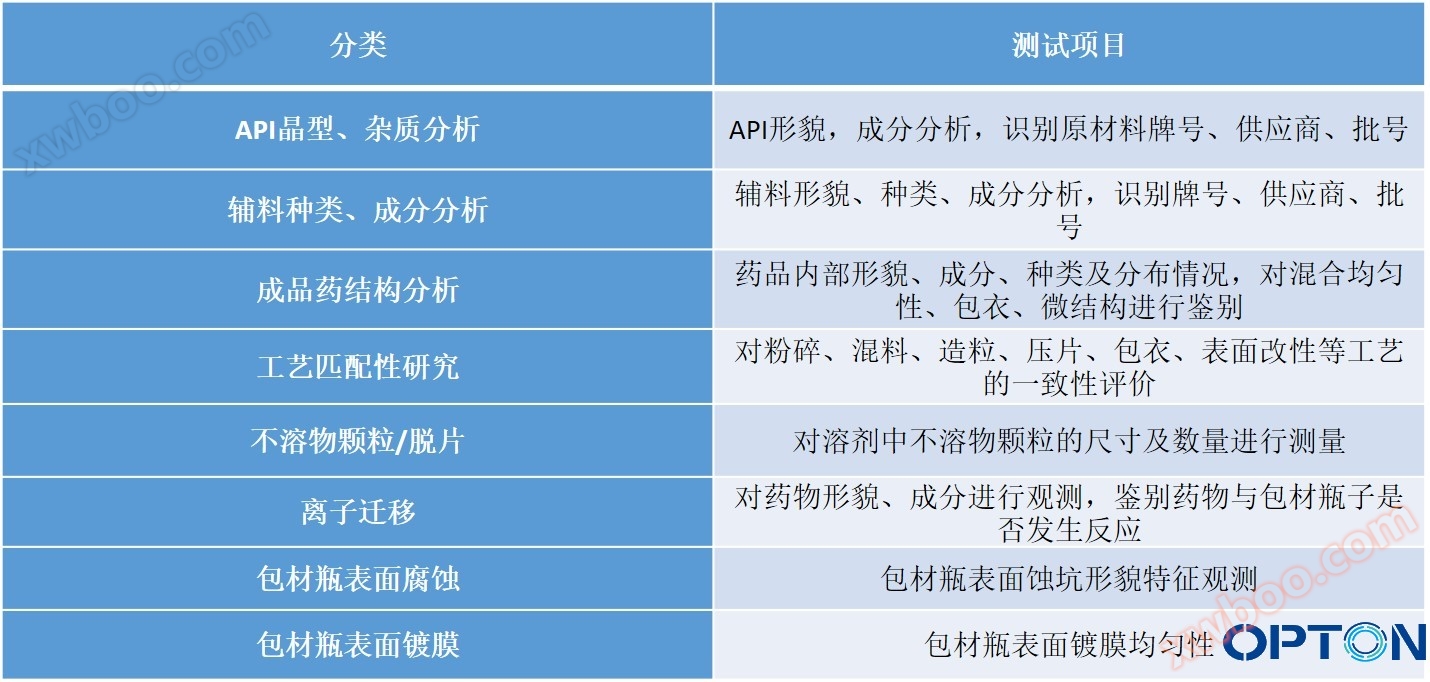
Chemical and Pharmaceutical Solutions - Test Project Summary
Drug consistency evaluation (tablets, targeted, sustained-release)
Example 1: Cross section of sustained-release drug particles - prediction of drug sustained-release performance
For water-insoluble sustained-release pellets, in order to achieve the desired drug release rate and ensure complete drug release, pore forming agents can be added to the sustained-release material to increase the porosity and permeability of the coating film or matrix, facilitating the regulation of drug release.
The SEM images of drug 1 # and 2 # are as follows. It can be seen that there are numerous pores on the surface and inside of sample 1 #, while the opposite is true for sample 2 #.
Example 2: Powder and tablet morphology
The same raw materials and different processes can produce medicinal powders with different morphologies and significant differences in performance.
To identify the uniformity of the mixing of raw materials inside the tablet, cross-sectional tablets can be prepared and directly observed from the morphology.
Example 3: Traceability and uniformity evaluation of drug raw materials

Example 4: Drug Consistency Evaluation (Tablets, Targeted, Extended Release)
Observation of finished drug packaging and thickness distribution using CT imaging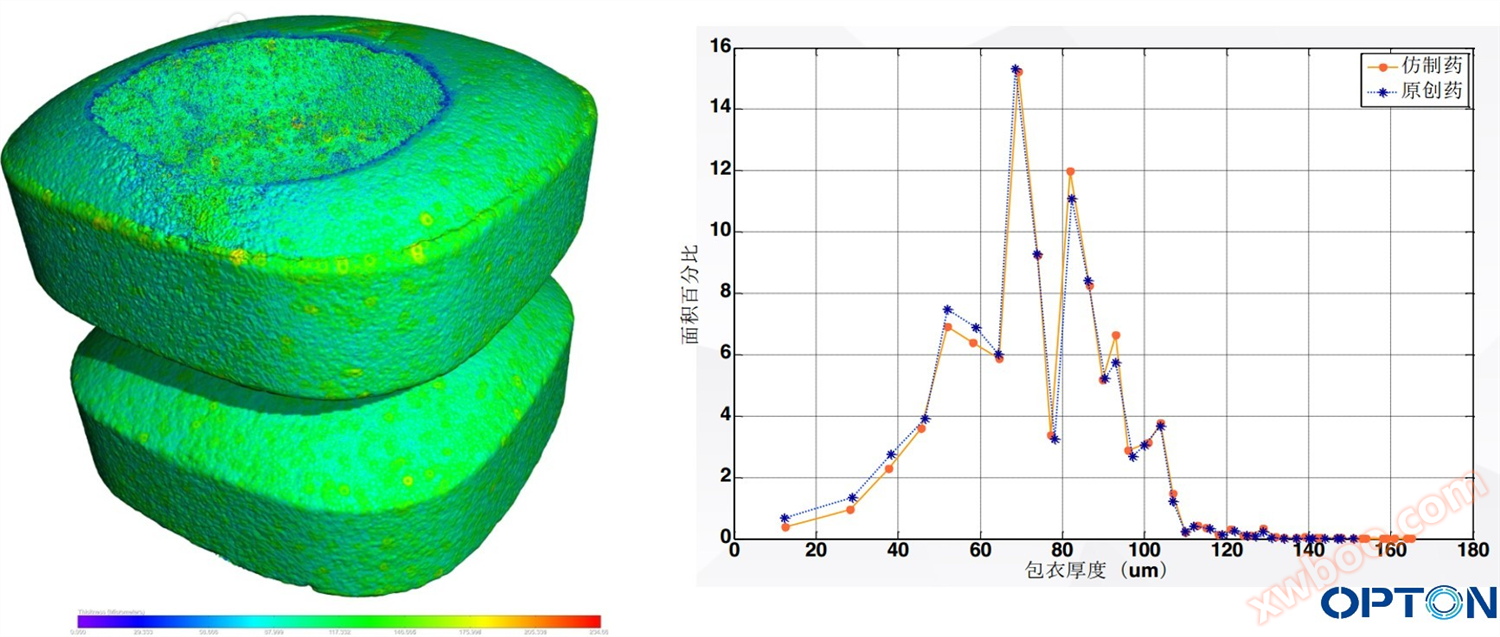
Example 5: Drug Consistency Evaluation (Tablets, Targeted, Extended Release)
Observation of Microcracks in Finished Drug Packaging Clothes Using CT Imaging

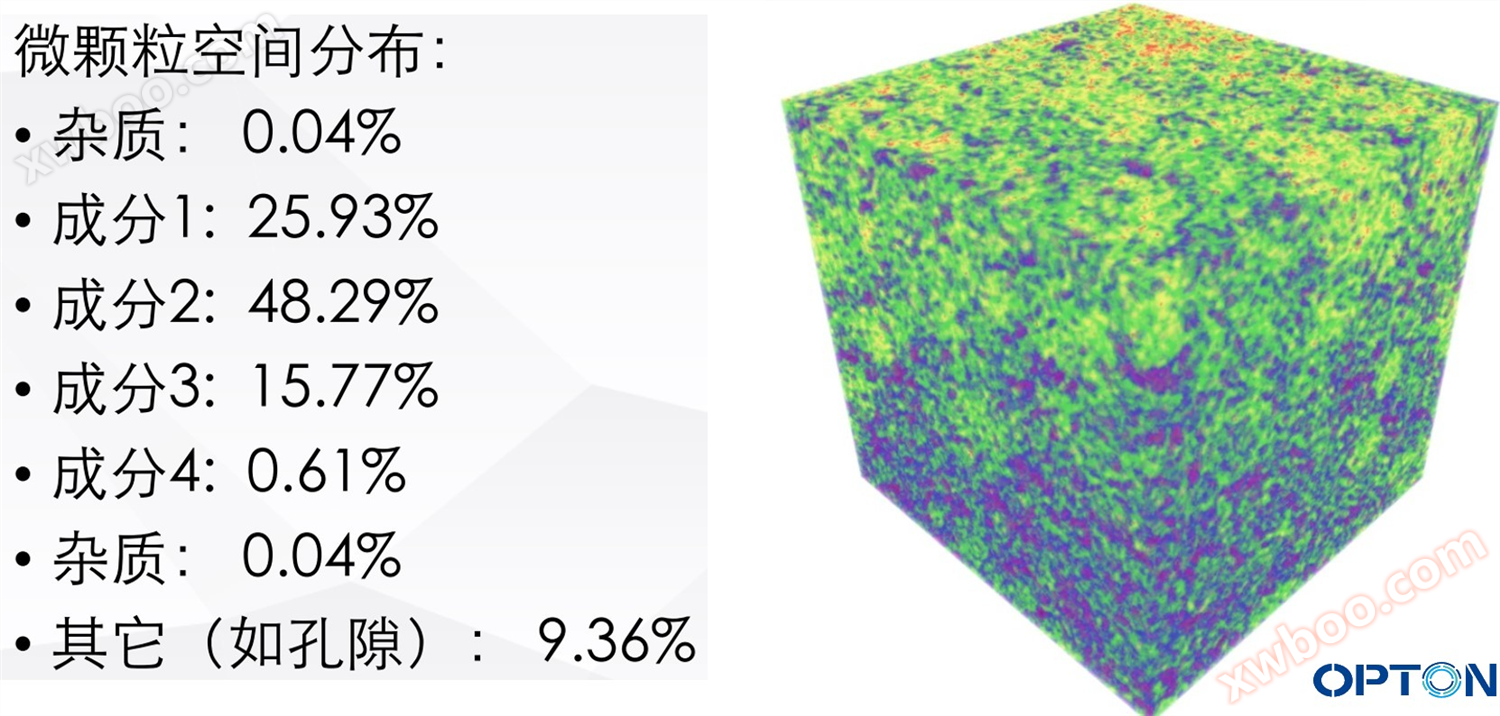
Example 6: Drug Consistency Evaluation (Tablets, Targeted, Extended Release)
Observation of finished drug particles and distribution using CT imaging
Compatibility of packaging materials
Example: Corrosion of packaging materials

